ISO 8 Hardwall Cleanroom with Control System & A/C
Our client manufactures medical device components, using a cleanroom for production to meet the Medical Device Directive.

Key facts
ISO 8 hardwall cleanroom
Custom-built 243m² cleanroom
Short lead time and quick installation
With control system & A/C
What did the client need?
Our client manufactures medical device components, using a cleanroom for production to meet the Medical Device Directive.
The company were expanding onto a new site, as demand for their product was growing; so they began looking for a cleanroom solution to meet their needs.
They were attracted by our short lead time and speed of installation, as this meant they could soon be capitalising on their investment.

How did the client benefit?
The quality of Connect 2 Cleanrooms’ Closed-Loop Control System, was advantageous. As our client needed to store audit trail data for their customers, the automated monitoring, recording and control of the particulate level within the cleanroom saves them a lot of time and effort.
The modular design of the cleanroom meant it could be treated as equipment as opposed to capital, which held a significant tax benefit. This combined with the lower cost per m² compared to a traditional build style of cleanroom made the modular approach very attractive.

How did we help the client?
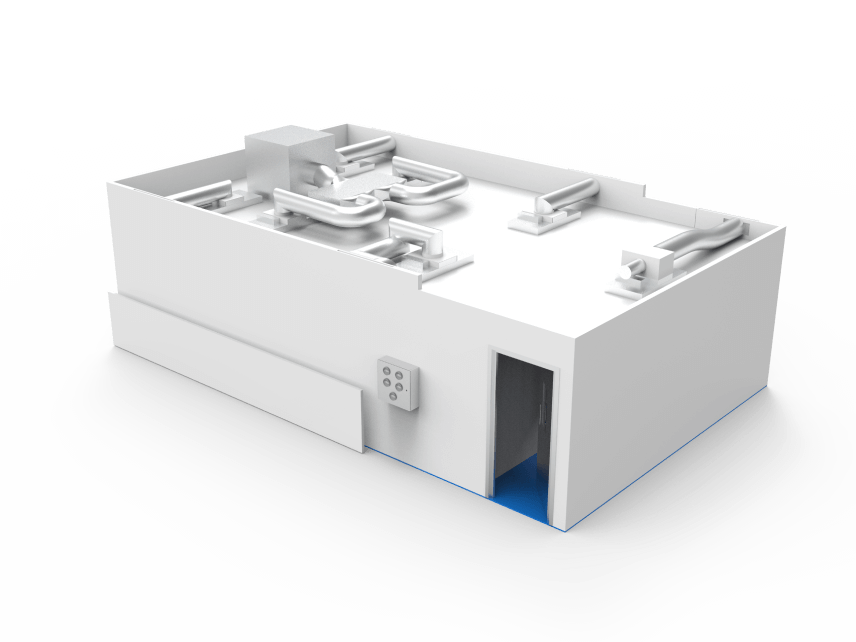
We accommodated production machinery by increasing the height of their cleanroom to 3.2m.
Cleanroom air conditioning units were built into the room for the purpose of comfort cooling.
The clear hardwall panels create a light and open working environment for production staff. The client’s logo was manifested on the clear hardwall panels for branding
The installation of the cleanroom followed the time schedule agreed and the client was impressed that it only took 4 days.
The validation report was “very thorough” and gave the client exactly what was needed to meet the regulatory requirements of the Medical Device Directive.


“I would be very happy to recommend Connect 2 Cleanrooms as I was impressed with their quality of service, build quality and performance.
“They have produced a nice and light environment for our production staff to work in.”

START A PROJECT WITH US
Our design and build specialists have experience working with customers in all kinds of industries on a global scale, achieving great results time and time again. We’d love to work with you as well!
REQUEST A QUOTE